Stainless Steel Pipe
Durable and corrosion-resistant stainless steel pipes for various applications.

Austenitic stainless steel is a type of stainless steel that is widely used in various industrial applications due to its excellent corrosion resistance and versatility.
Austenitic stainless steels are versatile and popular materials, known for their corrosion resistance, high-temperature strength, and suitability for the fabrication process.
A stable austenite structured steel contains about 18% Cr, 8% ~ 10% Ni and about 0.1% C. Chrome-nickel austenitic stainless steel including the most familiar stainless steel 18Cr-8Ni (TP304) and the series of high Cr-Ni steel developed by increase Cr, Ni contents and add Mo, Cu, Si, Nb, Ti and other elements. Austenitic stainless steel is characterized by non-magnetic, high plasticity and ductility but lower strength. It can be only strengthened via cold working (by adding S, Ca, Se, Te and etc, austenitic stainless steel can have excellent machinability).
Grades:304/L/H/LN, 316/L/H/LN/Ti/LMod, 310S/H, 317/L, 321/H, 347H/HFG
Specifications:
Austenitic steels are non-magnetic stainless steels that contain high levels of chromium and nickel and low levels of carbon. Known for their formability and resistance to corrosion, austenitic steels are the most widely used grade of stainless steel.
Features:
There is a fifth specialist type Precipitation Hardening Steels.
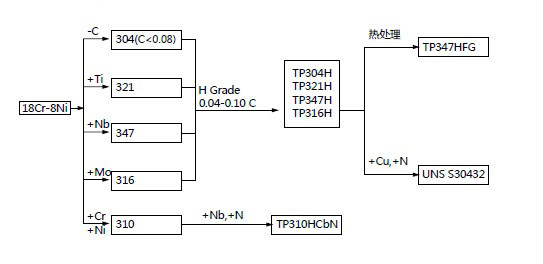
Development diagram of common grades of austenitic stainless steel
The fundamental difference between them is their crystaline structure. Steel is an alloy of iron and carbon. At normal temeperatures the atomic structure is that of a cube with an atom of steel at each corner and a single atom in the centre of that cube.
This is known as "ferritic" and, is by the way, magnetic.
When heated to about 900o C the structure changes and each face has an atom at its centre. This is the austenitic structure and is non-magnetic.
When ordinary steel cools down gradually it reverts to a ferritic structure. If you cool it fast, it will adopt another structure with the carbon atoms being arranged in one direction. This is martensitic steel and in its "as quenched" condition is hard but brittle and generally requires further treatment before it can be used.
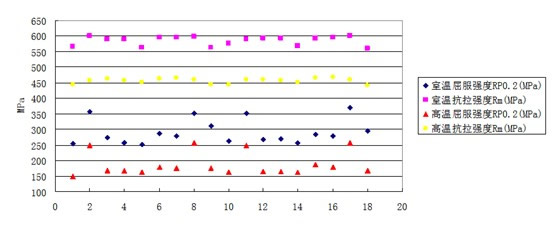
Strength Graph of 316L Mod at room and elevated temperature
| AISI grade | C max. | Si max. | Mn max. | Cr | Ni | Mo | Ti | Nb | Al | V |
| 301 | 0.15 | 1.00 | 2.00 | 16-18 | 6-8 | |||||
| 302 | 0.15 | 1.00 | 2.00 | 17-19 | 8-10 | |||||
| 304 | 0.08 | 1.00 | 2.00 | 17.5-20 | 8-10.5 | |||||
| 310 | 0.25 | 1.50 | 2.00 | 24-26 | 19-22 | |||||
| 316 | 0.08 | 1.00 | 2.00 | 16-18 | 10-14 | 2.0-3.0 | ||||
| 321 | 0.08 | 1.00 | 2.00 | 17-19 | 9-12 | 5 x %C min. | ||||
| 347 | 0.08 | 1.00 | 2.00 | 17-19 | 9-13 | 10 x %C min. | ||||
| E 1250 | 0.1 | 0.5 | 6.0 | 15.0 | 10.0 | 0.25 | ||||
| 20/25-Nb | 0.05 | 1.0 | 1.0 | 20.0 | 25.0 | 0.7 | ||||
| A 286 | 0.05 | 1.0 | 1.0 | 15.0 | 26.0 | 1.2 | ~1.9 | ~0.18 | ~0.25 | |
| 254SMO | 0.02 | 0.8 | 1.0 | 18.5-20.5 | 17.5-18.5 | 6-6.5 | ~1.9 | ~0.18 | ~0.25 | |
| AL-6XN | 0.03 | 1.0 | 2.0 | 20-22 | 23.5-25.5 | 6-7 |
Annealing
Annealing is a process of heating the steel to a temperature slightly above its recrystallisation temperature and allowing it to cool at an appropriate rate - generally slowly - causing the crystals to reform without the defects caused by "working" the steel.
Annealing can restore the ductility of the steel and its corrosion resistance characteristics.
Carbide Precipitation
When steel contains higher levels of carbon there is a tendency for it to combine with the chromium as it cools - between 900oC and 500oC forming chromium carbide. This reduces the amount of chromium available to form the passive layer and creates intergranular boundaries that are accessible to corrosive chemicals.
This can be overcome by using Low carbon variants of the steel (designated by "L", eg 304L or 316L).
However, the lower carbon levels reduce the steel's performance at elevated temperature. If resistance to carbide precipitation and strength at high temperature are required then the addition of Titanium may be the solution. There are a number of grades available in this form - eg 316Ti.
Creep Strength
Steels perform very differently at elevated temperatures than they do at ambient temperatures. When they are bent at ambient temperatures to below their yield point, they will spring back. At elevated temperatures, they begin to stretch, but very slowly. Some steels have better resistance to this phenomenon than others.
Grain Size
Steel is made up of a lattice of crystals of iron interwoven with atoms of other materials. These crystals are called grains.
The grain size is important because it affects the machining, hardness, strength, and corrosion resistance amongst other things.
Grain size can be determined both by the addition of other alloying elements and by the careful regulation of the heating and cooling processes involved in the production of the steel and by further heat treatment (annealing and quenching) following initial production, welding or "working" on the steel.
Intergranular Corrosion
The atoms in metals are arranged into crystals (or grains) which are aligned closely with each other. In certain conditions, corrosion can attack the grain boundaries rather than the crystals themselves.
When stainless steel containing a higher percentage of carbon is heated it the chromium can react with the carbon to form chromium carbide thereby depleting the passive layer of chromium that protects the surface.
Passive Layer
The passive layer is what makes stainless steel "stainless". It is a microscopically thin layer of chromium oxide that is impenetrable to oxygen, very hard, resists corrosion itself and is virtually transparent. This prevents oxygen and other corrosive materials reaching the iron and reacting with it.
Chromium reacts readily with oxygen with the result that should it become scratched it will repair itself providing there is free oxygen available.
Pitting Corrosion
This is a very localised form of corrosion that arises particularly in high chloride conditions such as the marine environment. An initial breach in the passive layer of the steel is not "repaired" by the reformation of chromium oxide. The steel beneath this breach continues to corrode often leaving no obvious signs other than a light surface staining (sometimes called "tea staining) on the surface but continuing to deepen and widen below the surface.
While localised it can result in the penetration of the entire cross section of the steel.
High contents of chromium, molybdenum and nitrogen increase the resistance to pitting corrosion. The degree of resistance can be calculated as %Chromium + 3.5 x %Molybdenum + 16 x %Nitrogen to give a Pitting Resistance Equivalent Number (PREN).
316 has a PERN of between 22.6 – 27.9. Some Duplex steels have PRENs over 40. The span of the numbers given in certain grades is the result of the specification for the quantities of the relevant chemical having max and min figures.
Precipitation hardening
Also known as age hardening, is a process used to increase tensile strength. The alloy is first raised to a temperature that a produces a single phase with all the solute atoms dissolved and evenly distributed. It is then rapidly quenched before reheating to a lower temperature and holding it at that temperature for a predetermined time. At this temperature, the precipitates can clump together in a uniform and distributed manner. It is essential that the right temperature and duration at this stage of the process is correct. If the temperature is held too long it will result in oversize clumps and reduce the strength of the alloy. This is known as "over aging".
Sensitisation
Sensitisation is the process of Carbide Precipitation - See above.
Sigma phase embrittlement.
Is phase change that occurs in some stainless steels when they are heated above about 540C. This results in a dramatic loss of toughness and can lead to brittle fractures.
Stabilisation
Stabilisation is the process of removing or protecting the steel from sensitisation - the danger of carbide precipitation which can lead to stress corrosion cracking (SCC).
There are two approaches commonly used. Low carbon variants can be used; they are inherently more stable but they perform less well at higher temperatures.
Alternatively, steel can be chemically stabilised by alloying it with titanium, niobium (sometimes still called columbium). Both of these readily form carbides thereby preserving the chromium.
However, this may not be sufficient to stabilise the steel should it be held within the carbide forming temperature band 425oC to 850oC. Should this occur during the fabrication process the problem can generally be reversed by annealing at a higher temperature.
Stress Corrosion Cracking (SCC)
Stress corrosion cracking occurs when chemicals attack the intergranular boundaries within an alloy. When the metal is subject to tensile stresses it can result in the sudden failure in generally ductile materials. Because the corrosion only occurs at the grain boundaries it is likely to go unnoticed since the metal will generally maintain an apparently normal surface appearance.
Work Hardening
Work hardening is a term applied to any work done on the steel at a temperature below the metal's recrystallisation temperature.
This work includes any type of squeezing, bending, cutting/shearing or drawing.
These processes cause distortions in the crystalline structure of the metal reducing their ability to move within the metal and makes it more resistant to further deformation.
Hardening can be an advantage or a disadvantage.
The crystalline structure can be restored by annealing.
Austenitic stainless steels are the most common and widely known types of stainless steels. They make up over 70 % of total stainless steel production. These steels contain around 16 % to 25 % chromium and sufficient nickel and/or manganese to retain an austenitic structure at all temperatures from cryogenic region to the melting point of the stainless steel. Austenitic stainless steels can also contain nitrogen in solution. Although nickel is the alloying element most commonly used to produce austenitic stainless steels, nitrogen can also be used to produce austenitic stainless steels. The austenitic stainless steels are more easily recognized because of their non magnetic properties. Austenitic steels are non magnetic since the face centered cubic structure of austenite is non magnetic. They are extremely formable and weldable, and they can be successfully used from cryogenic temperatures to the jet engines and red hot temperatures of furnaces.
Austenitic stainless steels are mainly segregated into the following two series
Besides the above two series there are super austenitic stainless steel grades which exhibit great resistance to chloride pitting and crevice corrosion because of high molybdenum content (> 6 %) and nitrogen additions. Higher nickel content ensures better resistance to stress-corrosion cracking than the stainless steels of the 300 series. The higher alloy content of super austenitic steels makes them more expensive.
The straight grades of stainless steel contain a maximum of 0.08 % carbon. In these grades, there is no requirement of minimum carbon in the specification.
The ‘L’ grades are used to provide extra corrosion resistance after welding. The letter ‘L’ after a stainless steel grade indicates low carbon (as in 304L). The carbon is kept to 0.03 % or under to avoid carbide precipitation. Carbon in steel, when heated to temperatures in what is called the critical range (430 deg C to 870 deg C) precipitates out, combines with the chromium and gathers on the grain boundaries. This deprives the steel of the chromium in solution and promotes corrosion adjacent to the grain boundaries. By controlling the amount of carbon, this is minimized. For weldability, the ‘L’ grades are used. However the ‘L’ grades are more expensive. In addition, carbon, at high temperatures imparts great physical strength.
The ‘H’ grades contain a minimum of 0.04 % carbon and a maximum of 0.10 % carbon and are designated by the letter ‘H’ after the steel grade. ‘H’ grades are primarily used at extreme temperatures as the higher carbon helps the material retain strength at extreme temperatures.
Austenitic stainless steels can also be classified into following three groups.
All austenitic stainless steels contain a small amount of ferrite. Conventional austenitic stainless steel grades may contain traces of delta ferrite, for improved weldability. Usually this amount of ferrite is not enough to attract a normal magnet. However, if the balance of elements in the steel favours the ferritic end of the spectrum, it is possible for the amount of ferrite to be sufficient to cause a significant magnetic response. Also, some types of stainless steels are deliberately balanced to have a significant amount of ferrite.
Properties and of stainless steels
Austenitic stainless steels are non magnetic and are not heat treatable. They cannot be hardened by heat treatment. However, they can be cold worked to improve hardness, strength and stress resistance. A solution anneal (heating within the range 1000 deg C to 1200 deg C followed by quenching or rapid cooling) restores the stainless steels original condition, including removal of alloy segregation and re-establishment of ductility after cold working. Stainless steels can be subjected to solution annealing. Due to the solution annealing the carbides, which may have precipitated (or moved) at the grain boundaries, are put back into solution (dispersed) into the matrix of the metal by the annealing process. ‘L’ grades are used where annealing after welding is impractical.
Austenitic stainless steels can be made soft enough (i.e. with yield strength of around 200 N/sq mm) to be easily formed by the same tools that work with carbon steel, but they can be made incredibly strong by cold work, up to yield strengths of over 2000 N/sq mm. Their austenitic (fcc, face centered cubic) structure is very tough and ductile down to absolute temperature. They also do not lose their strength at elevated temperatures as rapidly as ferritic (bcc, body centered cubic) iron base alloys.
Austenitic grades of stainless steels are the most common used grades, mainly because they provide very predictable level of corrosion resistance with excellent mechanical properties. The least corrosion resistant versions can withstand the normal corrosive attack of the everyday environment that people experience, while the most corrosion resistant grades can even withstand boiling seawater.
Austenitic stainless steels have good formability and weldability, as well as excellent toughness, particularly at low, or cryogenic, temperatures. Austenitic grades also have a low yield stress and relatively high tensile strength. They have excellent corrosion resistance and excellent high-temperature tensile and creep strength.
Austenitic stainless steels are not very strong materials. Typically their 0.2 % proof stress is about 250 N/sq mm and the tensile strength between 500 and 600 N/sq mm, showing that these steels have substantial capacity for work hardening, which makes working more difficult than in the case of mild steel. However, austenitic stainless steels possess very good ductility with elongations of about 50 % in tensile tests.
Austenitic stainless steels are also highly resistant to high temperature oxidation because of the protective surface film, but the usual grades have low strengths at elevated temperatures. Those steels stabilized with Ti and Nb, grades 321 and 347, can be heat treated to produce a fine dispersion of TiC or NbC which interacts with dislocations generated during creep. One of the most commonly used alloys is 25Cr20Ni with additions of titanium or niobium which possesses good creep strength at temperatures as high as 700 deg C.
Austenitic stainless steels are ductile over a wide temperature range, from cryogenic to creep temperatures. They do not display brittle fracture. Their tensile strength is high at low temperatures. They can be work hardened to high levels of strength by cold forming.
Austenitic stainless steels are less resistant to cyclic oxidation than are ferritic grades because their greater thermal expansion coefficient tends to cause the protective oxide coating to spall. They can experience stress corrosion cracking (SCC) if used in an environment to which they have insufficient corrosion resistance. The fatigue endurance limit is only about 30 % of the tensile strength (vs. 50 % – 60 % for ferritic stainless steels). This, combined with their high thermal expansion co efficient, makes them especially susceptible to thermal fatigue. However, the risks of these limitations can be avoidable by taking special precautions.
The salient feature of austenitic stainless steels is that as chromium and molybdenum contents are increased to increase specific properties, usually corrosion resistance, nickel or other austenitic stabilizers must be added if the austenitic structure is to be preserved.
The tensile properties in the annealed state not surprisingly relate well to composition. The 0.2 % yield strength applies to the austenitic stainless steels.
Austenitic stainless steels have many advantages from a metallurgical point of view. Their properties include good to excellent corrosion resistance. They can be work hardened. They can be easily machined and fabricated to tight tolerances. They have smooth surface finish that can be easily cleaned and sterilized. They are temperature resistant from cryogenic to high heat temperatures.
Austenitic stainless steels are classified in the 200 and 300 series, with 16% to 30% chromium and 2% to 20% nickel for enhanced surface quality, formability, increased corrosion and wear resistance. Austenitic stainless steels are non-hardenable by heat treating. These steels are the most popular grades of stainless produced due to their excellent formability and corrosion resistance. All austenitic steels are nonmagnetic in the annealed condition. Depending on the composition, some austenitics do become somewhat magnetic when cold worked. Austenitics are used for automotive trim, cookware, food and beverage equipment, processing equipment, and a variety of industrial applications.
Corrosion resistance in stainless steels is primarily determined by chromium content. Austenitic stainless steels, as a class, have excellent corrosion resistance and those with molybdenum additions have improved pitting resistance. The nickel content in austenitic stainless steels helps to reduce the rate of corrosion, particularly in acid environments. Austenitic grades, however, are susceptible to chloride stress corrosion cracking (SCC) and are not recommended for service that combines tensile stress and the presence of chlorides, even at moderate temperatures. Higher carbon austenitic grades can be susceptible to intergranular corrosion after certain high temperature exposures, including welding. For applications that require welding, a post-weld heat treatment or selection of a low carbon or stabilized grade such as Type 304L, 316L and 321, is recommended.
304 and 304L (standard grade):
309 and 310 (high chrome and nickel grades):
318 and 316L (high moly content grades):
321 and 316Ti ("stabilized" grades):
200 Series (low nickel grades):
Austenitic steels are non-magnetic stainless steels that contain high levels of chromium and nickel and low levels of carbon. Known for their formability and resistance to corrosion, austenitic steels are the most widely used grade of stainless steel.
Defining Characteristics Ferritic steels have a body-centered cubic (BCC) grain structure, but the austenitic range of stainless steels are defined by their face-centered cubic (FCC) crystal structure, which has one atom at each corner of the cube and one in the middle of each face. This grain structure forms when a sufficient quantity of nickel is added to the alloy—8 to 10 percent in a standard 18 percent chromium alloy.
In addition to being non-magnetic, austenitic stainless steels are not heat treatable. They can be cold worked to improve hardness, strength, and stress resistance, however. A solution anneals heated to 1045° C followed by quenching or rapid cooling will restore the alloy's original condition, including removing alloy segregation and re-establishing ductility after cold working.
Nickel-based austenitic steels are classified as 300 series. The most common of these is grade 304, which typically contains 18 percent chromium and 8 percent nickel.
Eight percent is the minimum amount of nickel that can be added to a stainless steel containing 18 percent chromium in order to completely convert all the ferrite to austenite. Molybdenum can also be added to a level of about 2 percent for grade 316 to improve corrosion resistance.
Although nickel is the alloying element most commonly used to produce austenitic steels, nitrogen offers another possibility. Stainless steels with a low nickel and high nitrogen content are classified as 200 series. Because it is a gas, however, only limited amounts of nitrogen can be added before deleterious effects arise, including the formation of nitrides and gas porosity that weaken the alloy.
The addition of manganese, also an austenite former, combined with the inclusion of nitrogen allows for greater amounts of the gas to be added. As a result, these two elements, along with copper—which also has austenite-forming properties—are often used to replace nickel in 200 series stainless steels.
The 200 series—also referred to as chromium-manganese (CrMn) stainless steels—were developed in the 1940s and 1950s when nickel was in short supply and prices were high. It is now considered a cost-effective substitute for 300 series stainless steels that can provide an additional benefit of improved yield strength.
Straight grades of austenitic stainless steels have a maximum carbon content of 0.08 percent. Low carbon grades or "L" grades contain a maximum carbon content of 0.03 percent in order to avoid carbide precipitation.
Austenitic steels are non-magnetic in the annealed condition, although they can become slightly magnetic when cold worked. They have good formability and weldability, as well as excellent toughness, particularly at low or cryogenic temperatures. Austenitic grades also have a low yield stress and relatively high tensile strength.
While austenitic steels are more expensive than ferritic stainless steels, they are generally more durable and corrosion resistant.
Nickel which stabilizes the austenitic structure of these steels restricts their widespread usage since nickel increases the costs of these stainless steels.
Other steels can offer similar performance at lower cost and are preferred in certain applications, for example ASTM A387 is used in pressure vessels but is a low-alloy carbon steel with a chromium content of 0.5 % to 9 %. Low-carbon versions, for example 316L or 304L, are used to avoid corrosion problems caused by welding. Grade 316LVM is preferred where biocompatibility is required (such as body implants and piercings).
Austenitic grades of stainless steels are the most commonly used grades, mainly because they provide very predictable level of corrosion resistance with excellent mechanical properties. Using them wisely can save the designer of a product significant cost. These steels are user friendly metal alloy with life cycle cost of fully manufactured products lower than many other materials.
Austenitic stainless steels are those steels which are commonly used for stainless application. Some of the applications for austenitic stainless steel include the following.
Super austenitic stainless steels contain high levels of chromium and higher levels of nickel together with additions of molybdenum and nitrogen. The result is a series of austenites, stronger than conventional 300 series stainless and with superior resistance to pitting, crevice corrosion, and stress corrosion cracking.
Super austenitic stainless steel is defined as Cr-Ni stainless steel with a Pitting Resistance Equivalent Number (PREN=[Cr]+3.3[Mo]+16[N])≥40%. It has better resistance to chloride pitting and crevice corrosion than Cr-Ni austenitic stainless steel with Mo >4% in highly corrosive media containing Cl-
Grades:
Features:
Specifications:
Machinability
Work hardening produces hard surfaces and hard chips , which in turn lead to notch wear. It also creates adhesion and produces built-up edge (BUE). It has a relative machinability of 60%. The hardening condition can tear coating and substrate material from the edge, resulting in chipping and bad surface finish. Austenite produces tough, long, continuous chips, which are difficult to break. Adding S improves machinability, but results in lowered resistance to corrosion.
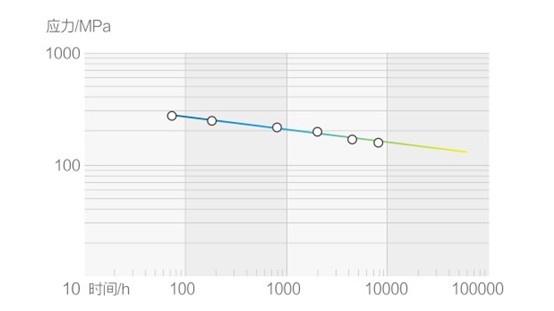
Rupture Strength Curve of S30432 Ultra-Supercritical Boiler Tube
Use sharp edges with a positive geometry.
Cut under the work hardened layer.
Keep cutting depth constant. Generates a lot of heat when machined.

The application fields are very wide, mainly nuclear power, oil refining industry, atmospheric and vacuum equipment, catalytic cracking equipment, hydrocracking, hydrotreating equipment; Vinyl chloride production units, formaldehyde synthesis reactors, acetic acid and other organic acid production units in the petrochemical and chemical industry; The oil and gas industry; Pulp and paper industry; Urea industry and phosphate fertilizer industry in fertilizer industry; Seawater environment; Energy and environmental protection industry; Salt and chemical equipment for light industry and food industry, equipment for food and pharmaceutical industry, etc.
In order to solve the cumbersome and difficult to remember stainless steel grades, improve the practicability of the brand representation, and the contrast with the international standard grades, China has formulated the "Universal Code System for Steel and Alloy Grades", such as 06Cr19Ni10, corresponding to 304. Different grades of stainless steel have different ingredients, but they all have a national standard. The standards of each country are also different.
| No | China,GB | Japan | American | Korea | EUR | Indai | Australia | Tai Wan | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Old | New(07.10) | JIS | ASTM | UNS | KS | BS EN | IS | IS | CNS | |
| Austenitic stainless steel | ||||||||||
| 1 | 1Cr17Mn6Ni5N | 12Cr17Mn6Ni5N | SUS201 | 201 | S20100 | STS201 | 1.4372 | 10Cr17Mn6Ni4N20 | 201-2 | 201 |
| 2 | 1Cr18Mn8Ni5N | 12Cr18Mn9Ni5N | SUS202 | 202 | S20200 | STS202 | 1.4373 | - | 202 | |
| 3 | 1Cr17Ni7 | 12Cr17Ni7 | SUS301 | 301 | S30100 | STS301 | 1.4319 | 10Cr17Ni7 | 301 | 301 |
| 4 | 0Cr18Ni9 | 06Cr19Ni10 | SUS304 | 304 | S30400 | STS304 | 1.4301 | 07Cr18Ni9 | 304 | 304 |
| 5 | 00Cr19Ni10 | 022Cr19Ni10 | SUS304L | 304L | S30403 | STS304L | 1.4306 | 02Cr18Ni11 | 304L | 304L |
| 6 | 0Cr19Ni9N | 06Cr19Ni10N | SUS304N1 | 304N | S30451 | STS304N1 | 1.4315 | - | 304N1 | 304N1 |
| 7 | 0Cr19Ni10NbN | 06Cr19Ni9NbN | SUS304N2 | XM21 | S30452 | STS304N2 | - | - | 304N2 | 304N2 |
| 8 | 00Cr18Ni10N | 022Cr19Ni10N | SUS304LN | 304LN | S30453 | STS304LN | - | - | 304LN | 304LN |
| 9 | 1Cr18Ni12 | 10Cr18Ni12 | SUS305 | 305 | S30500 | STS305 | 1.4303 | - | 305 | 305 |
| 10 | 0Cr23Ni13 | 06Cr23Ni13 | SUS309S | 309S | S30908 | STS309S | 1.4833 | - | 309S | 309S |
| 11 | 0Cr25Ni20 | 06Cr25Ni20 | SUS310S | 310S | S31008 | STS310S | 1.4845 | - | 310S | 310S |
| 12 | 0Cr17Ni12Mo2 | 06Cr17Ni12Mo2 | SUS316 | 316 | S31600 | STS316 | 1.4401 | 04Cr17Ni12Mo2 | 316 | 316 |
| 13 | 0Cr18Ni12Mo3Ti | 06Cr17Ni12Mo2Ti | SUS316Ti | 316Ti | S31635 | - | 1.4571 | 04Cr17Ni12MoTi20 | 316Ti | 316Ti |
| 14 | 00Cr17Ni14Mo2 | 022Cr17Ni12Mo2 | SUS316L | 316L | S31603 | STS316L | 1.4404 | -02Cr17Ni12Mo2 | 316L | 316L |
| 15 | 0Cr17Ni12Mo2N | 06Cr17Ni12Mo2N | SUS316N | 316N | S31651 | STS316N | - | - | 316N | 316N |
| 16 | 00Cr17Ni13Mo2N | 022Cr17Ni13Mo2N | SUS316LN | 316LN | S31653 | STS316LN | 1.4429 | - | 316LN | 316LN |
| 17 | 0Cr18Ni12Mo2Cu2 | 06Cr18Ni12Mo2Cu2 | SUS316J1 | - | - | STS316J1 | - | - | 316J1 | 316J1 |
| 18 | 00Cr18Ni14Mo2Cu2 | 022Cr18Ni14Mo2Cu2 | SUS316J1L | - | - | STS316J1L | - | - | - | 316J1L |
| 19 | 0Cr19Ni13Mo3 | 06Cr19Ni13Mo3 | SUS317 | 317 | S31700 | STS317 | - | - | 317 | 317 |
| 20 | 00Cr19Ni13Mo3 | 022Cr19Ni13Mo3 | SUS317L | 317L | S31703 | STS317L | 1.4438 | - | 317L | 317L |
| 21 | 0Cr18Ni10Ti | 06Cr18Ni11Ti | SUS321 | 321 | S32100 | STS321 | 1.4541 | 04Cr18Ni10Ti20 | 321 | 321 |
| 22 | 0Cr18Ni11Nb | 06Cr18Ni11Nb | SUS347 | 347 | S34700 | STS347 | 1.455 | 04Cr18Ni10Nb40 | 347 | 347 |
| Austenitic ferritic stainless steel (duplex stainless steel) | ||||||||||
| 23 | 0Cr26Ni5Mo2 | - | SUS329J1 | 329 | S32900 | STS329J1 | 1.4477 | - | 329J1 | 329J1 |
| 24 | 00Cr18Ni5Mo3Si2 | 022Cr19Ni5Mo3Si2N | SUS329J3L | - | S31803 | STS329J3L | 1.4462 | - | 329J3L | 329J3L |
| 0Cr18Ni10Ti Iron type stainless steel | ||||||||||
| 25 | 0Crl3Al | 06Crl3Al | SUS405 | 405 | S40500 | STS405 | 1.4002 | 04Cr13 | 405 | 405 |
| 26 | - | 022Cr11Ti | SUH409 | 409 | S40900 | STS409 | 1.4512 | - | 409L | 409L |
| 27 | 00Cr12 | 022Cr12 | SUS410L | - | - | STS410L | - | - | 410L | 410L |
| 28 | 1Cr17 | 10Cr17 | SUS430 | 430 | S43000 | STS430 | 1.4016 | 05Cr17 | 430 | 430 |
| 29 | 1Cr17Mo | 10Cr17Mo | SUS434 | 434 | S43400 | STS434 | 1.4113 | - | 434 | 434 |
| 30 | - | 022Cr18NbTi | - | - | S43940 | - | 1.4509 | - | 439 | 439 |
| 31 | 00Cr18Mo2 | 019Cr19Mo2NbTi | SUS444 | 444 | S44400 | STS444 | 1.4521 | - | 444 | 444 |
| Martensitic stainless steel | ||||||||||
| 32 | 1Cr12 | 12Cr12 | SUS403 | 403 | S40300 | STS403 | - | - | 403 | 403 |
| 33 | 1Cr13 | 12Cr13 | SUS410 | 410 | S41000 | STS410 | 1.4006 | 12Cr13 | 410 | 410 |
| 34 | 2Cr13 | 20Cr13 | SUS420J1 | 420 | S42000 | STS420J1 | 1.4021 | 20Cr13 | 420 | 420J1 |
| 35 | 3Cr13 | 30Cr13 | SUS420J2 | - | - | STS420J2 | 1.4028 | 30 Cr13 | 420J2 | 420J2 |
| 36 | 7Cr17 | 68Cr17 | SUS440A | 440A | S44002 | STS440A | - | - | 440A | 440A |
In the production process, because the actual size is difficult to meet the nominal pipe size requirement, that is, it is often larger or smaller than the nominal size, so the standard stipulates that there is a difference between the actual size and the nominal size of the stainless steel pipe.
A positive difference is called a positive deviation, and a negative difference is called a negative deviation.
Outside diameter (OD), wall thickness and length are three basic parameters in the process of manufacture and distribution for stainless steel tube & pipe, and these parameters have been standardized.
Outer diameter and wall thickness are particularly important, they are not only the basic support for meeting the design requirements of various types of applications such as strength, stiffness and fluid delivery, but also have a decisive influence on the subsequent processing, therefore, standards for stainless steel tube & pipe diameter and wall thickness of the manufacturing accuracy (tolerance) should been standardized.
A. Nominal Pipe size: It is the nominal size specified in standards such as ASME B36.10m and ASME B36.19m. It is the ideal size that users and manufacturers hope to obtain, and it is also the order size specified in the contract.
B. Actual Pipe size: It is the actual size obtained during the production process. This size is often larger or smaller than the nominal size. The phenomenon of being larger or smaller than the nominal size is called deviation.
European stainless steel tube & pipe standards specify 4 classes outer diameter and wall thickness in percentage or absolute value on the basis of nominal dimension.
Generally, larger diameter or heavy wall thickness stainless steel pipe use percentage, small diameter or thin wall thickness stainless steel tube use absolute value.
European stainless steel tube & pipe standards specify 4 classes outer diameter and wall thickness in percentage or absolute value on the basis of nominal dimension.
Generally, larger diameter or heavy wall thickness stainless steel pipe use percentage, small diameter or thin wall thickness stainless steel tube use absolute value.
| Standard | Manufacturing Process & Type | Outer Diameter Tolerance | Wall Thickness Tolerance | Others | |
|---|---|---|---|---|---|
| OD (Mm) | Class | ||||
| EN10216-5 | Hot Finished Seamless Tube | 219.1-610 | D1 | +22.5%T,-15%T | T/D≤0.05 |
| T1 | T/D≤0.09 | ||||
| T2 | T/D>0.09 | ||||
| 30-219.1 | D2 | T1 | |||
| T2 | |||||
| Cold Finished Seamless Tubes | ≤219.1 | D3 | T3 | ||
| D4 | T4 | Agreement | |||
| EN10297-2 | Hot Finished Seamless Tube | D1 | T1 | ||
| D2 | T2 | Agreement | |||
| Cold Finished Seamless Tubes | D3 | T3 | |||
| D4 | T4 | Agreement | |||
| EN10217-7 | Welded Tubes | >168.3 | D2 | T3 | |
| ≤168.3 | D3 | T3 | |||
| D4 | T3 | Agreement | |||
| EN10296-2 | Welded Tubes | >168.3 | D2 | T3 | |
| ≤168.3 | D3 | T3 | |||
| ≤114.3 | D4 | T3 | Agreement | ||
| EN10312 | Welded Tubes | Series 1 | D4 | T3 | T=0.6-2 Mm |
| Series 2 | D3 – D4 | T3 – T4 | T=1-3 Mm | ||
From D1 to D4 class for outer diameter, and T1 to T4 for wall thickness tolerance comply to DIN EN ISO 1127 standard, stainless steel tubes – Dimensions, tolerances and conventional masses per unit length.
| Outside Diameter | Wall Thickness | ||
|---|---|---|---|
| Class | Tolerance | Class | Tolerance |
| D1 | ±1.5%, Or ±0.75 Mm Min. | T1 | ±15%, Or ±0.6 Mm Min. |
| D2 | ±1.0%, Or ±0.5 Mm Min. | T2 | ±12.5%, Or ±0.4 Mm Min. |
| D3 | ±0.75%, Or. ±0.3 Mm Min. | T3 | ±10%, Or ±0.2 Mm Min. |
| D4 | ±0.50%, Or ±0.1 Mm Min. | T4 | ±7.5%, Or ±0.15 Mm Min. |
| T5 | +/- 5.0 %, Or +/- 0.10 Mm Min. | ||
The tolerances on outside diameter and thickness from above table, greater value shall be selected.
EN 10312 has specific values in table series 1 and series 2.
America standard has two standard rules to specify outside diameter and thickness, ASTM A999 / A999M and A1016 / A1016M.
| ASTM A999 | ASTM A1016 |
|
|
But A511 for seamless stainless steel mechanical tubing and A554 welded stainless steel mechanical tubing not conform to above standards, they have separate outside diameter and thickness tolerances.
| Standard | Manufacturing & delivery condition | Outside Diameter OD / mm |
OD tolerance mm | Thickness T / mm |
T tolerance mm | |
|---|---|---|---|---|---|---|
| Minimum | Average | |||||
| ASTM A1016 A1016M |
Hot drawn seamless tubes | <=100 | +0.4, -0.8 | <=2.4 | +0.4t, 0 | |
| 100 – 200 | +0.4, -1.2 | 2.4 – 3.8 | +0.35t, 0 | |||
| 200 – 225 | +0.4, -1.6 | 3.8 – 4.6 | +0.33t, 0 | |||
| >=4.6 | +0.28t, 0 | |||||
| Cold drawn seamless tubes | <25 | +0.1, -0.11 | +0.20t, 0 | +/-0.10t | ||
| 25 – 40 | ||||||
| 40 – 50 | +/-0.2 | +0.22t, 0 | +/-0.10t | |||
| 50 – 65 | +/-0.25 | |||||
| 65 – 75 | +/-0.3 | |||||
| 75 – 100 | +/-0.38 | |||||
| 100 – 200 | +/-0.38, -0.04 | |||||
| 200 – 250 | +/-0.38, -1.14 | |||||
| Welded tubes | 40 – 50 | +/-0.2 | +0.18t, 0 | +/-0.10t | ||
| 50 – 65 | +/-0.25 | |||||
| 65 – 75 | +/-0.3 | |||||
| 75 – 100 | +/-0.38 | |||||
| 100 – 200 | +/-0.38, -0.04 | |||||
| 200 – 250 | +/-0.38, -1.14 | |||||
| ASTM A999 / M | Seamless tubes and welded tubes | <48.3 | +0.4, -0.8 | -0.125t | ||
| ASTM A312 / M | Seamless tubes and welded tubes | 48.3-114.3 | +/-0.8 | OD=10.3-73 | +0.20 t, -125t | |
| 168.3-219.1 | +1.6, -0.8 | t/OD<=5% OD=88.9-457.2 | +0.225 t, -125t | |||
| 219.1-457.2 | +2.4, -0.8 | t/OD>5% OD=88.9-457.2 | +0.15 t, -125t | |||
| Welded tubes | 508-660 | +3.2, -0.8 | OD>=508 | +0.175 t,-0.125 t | ||
| 711-864 | +4.0, -0.8 | t/OD<=5%, OD>=508 | +0.225 t,-0.125 t | |||
| Seamless tubes | 914-1209 | +4.8, -0.8 | t/OD>5%, OD>=508 | +0.15 t, -125t | ||
| ASTM A409 / M | Welded tubes | +/-0.2% | T<4.8 | |||
| +/-0.4% | t>=4.8 | |||||
| ASTM A358 / M | Welded tubes | +/-0.50% | ||||
| ASTM A511 | Seamless tubing | <=12.7 | +/-0.1 | +/-0.15t | ||
| 12.7-38.1 | +/-0.2 | +/-0.10t | ||||
| 38.1-88.9 | +/-0.3 | |||||
| 88.9-139.7 | +/-0.4 | |||||
| 139.7-203.1 | +/-0.8 | |||||
| 203.1-220 | +/-1.1 | |||||
| 220-325 | +/-1.6 | |||||
American standard outside diameter tolerance mainly used to represent the absolute value, over and below tolerances are often asymmetric, SS tube and pipe in American standard average tolerance can reach even higher than D4 class in EN ISO 1127 standard.
Wall thickness tolerance should be +/-10%t or better, and average thickness tolerance should be ( +/-20 – 22%t, 0 ), pipeline pipe has a larger tolerance, hot finished seamless pipe largest.
China has more quantity standards compared to European standards, but less than american standards, SS tube and pipe standards are similar with EU standards in system, and absorb favorable experience in USA specifications.
| Specification | Manufacturing | O.D(Mm) | O.D Tolerance | W.T(Mm) | W.T Tolerance | |
|---|---|---|---|---|---|---|
| GB13296 | Cold Rolled Seamless Pipe | 6-30 | ±0.15/-0.2 | 1-3 | +20% | -0% |
| >30-50 | ±0.3 | >3 | +22% | -0% | ||
| >50 | ±0.75% | Also Average Wall | ||||
| GB/T14976 | Cold Rolled Seamless Tube | 6-10 | ±0.15 | 1-3 | +12.5% | -12.5% |
| 10-30 | ±0.2 | >3 | +12.5% | -10% | ||
| 30-50 | ±0.3 | Average Wall, +22% | ||||
| >50 | ±0.8% | |||||
Stainless steel tube & pipe wall thickness tolerance are the same as the general accuracy of American ASTM / ASME and EU standards, but slightly lower than the high requirements of the EU standards.
Stainless steel tube & pipe outer diameter tolerances are mostly lower than American standards, and approaching the EU standards. it is more reasonable that the American standard specify the OD tolerance is tight, compared to thickness tolerance.
China GB standards and EU standard have no weight tolerance specified, seamless tube & pipe in GB standard delivery goods as actual weight or theoretical weight, welded tube & pipe as theoretical weight or actual weight.
Out of roundness, sometimes referred to as ovality is tube or pipe on the same cross-section of the outer diameter, roundness or ovality is difference between the maximum and minimum dimensions of outside diameter, is carefully measured the high and low points at any one section of the tube or pipe.
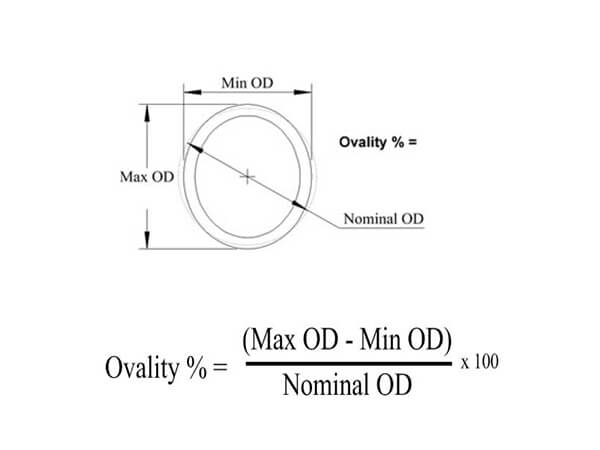
Concentricity or eccentricity refers to tube & pipe wall thickness variations, if need a high precision in tube tolerance, or fit into other tube or parts, which need a good concentricity.
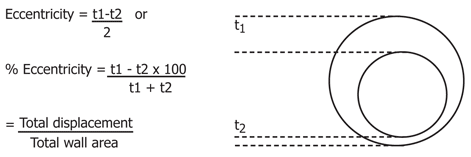
Concentricity be contained wall thickness tolerance specified in EU standard, and just specified in ASTM A1016/M OD≥50 mm and t≥5.6 mm thick wall stainless steel tube the same cross section thickness varieties.
For seamless tube,
WTmax – WTmin ≤ ±10% (WTmax + WTmin) / 2
For welded tube,
WTmax – WTmin ≤ 5% (WTmax + WTmin) / 2
Standard pipes and tubes are supplied straightened to the eye: for special applications the permissible deviation from the straight line may be agreed between purchaser and tube manufacturer; the maximum permissible deviation from the straight line related to the length of measurement L is to be indicated, e.g. 1mm/1000mm.
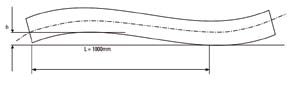
| OD Mm ≤ | OD Inches ≤ | Wall | Straightness Commercial | FT Achievable Mm |
|---|---|---|---|---|
| 15.9 | 0.625 | All Sizes | 1 In 600 | 1 In 2000 |
| 25.4 | 1.00 | 2% Of OD Or Heavier | 1 In 600 | 1 In 1500 |
| 25.4 | 1.00 | Less Than 2% Of OD | 1 In 400 | 1 In 1000 |
Delivery length is also called the length required by the user or the length of the order. The standard has the following regulations on delivery length:
A. Normal length / Random Length (also called non-fixed-length length): Any stainless steel tube whose length is within the length range specified by the standard and has no fixed length requirement is called normal length. For example, the structural stainless steel pipe standard stipulates: hot-rolled (extruded, expanded) steel pipe 3000mm - 12000mm; cold drawn (rolled) steel pipe 2000mm - 10500mm.
B. Fixed Length: The fixed-length should be within the usual length range, which is a certain fixed-length dimension required in the contract. However, it is impossible to cut out the absolute fixed-length length in actual operation, so the standard stipulates the allowable positive deviation value for the fixed-length length.

The yield rate of production of fixed-length pipes is much lower than that of normal-length pipes, and it is reasonable for manufacturers to request price increases. The rate of price increase varies from company to company. Generally, the price increase is about 10% on the basis of the base price.
C. Length of double ruler: The length of the double ruler should be within the usual length. The contract should indicate the length of the single ruler and the multiples of the total length (for example, 3000mm×3, which is 3 multiples of 3000mm, and the total length is 9000mm). In actual operation, the allowable positive deviation of 20mm should be added to the total length, plus a margin for the incision of each single ruler length. Take the structural pipe as an example, the allowance for cutting is stipulated: 5-10mm for outer diameter ≤159mm; 10-15mm for outer diameter >159mm.
If there is no double-length deviation and cutting allowance in the standard, it should be negotiated by the supplier and the buyer and indicated in the contract. The double-length scale is the same as the fixed-length length, which will bring about a substantial reduction in the yield rate of the manufacturer. Therefore, it is reasonable for the manufacturer to raise the price, and the price increase is basically the same as the fixed-length length.
D. Range length: The range length of the stainless steel pipe is within the usual length range. When the user requires a fixed range length, it must be indicated in the contract.
For example: Usually the length is 3000-12000mm, and the range cut-to-length length is 6000-8000mm or 8000-10000mm.
It can be seen that the range length is looser than the fixed-length and double-length length, but it is much stricter than the usual length, which will also reduce the yield of production enterprises. Therefore, it is reasonable for the manufacturer to raise the price, and the price increase is generally about 4% above the base price.
Stainless steel pipe (tube) has excellent characteristics of corrosion resistance and smooth finishing. Stainless steel pipe (tube) is commonly used in demanding equipment like automobiles, food processing, water treatment facilities, oil and gas processing, refinery and petrochemicals, breweries and energy industries.
The stainless steel tubing that is supplied by SunnySteel can used in a variety of industries, including:
Considering the importance of outside and inside surface of stainless steel tubes for fluid power industry, Our mills are providing tubes that are free from scale, rust, seams, laps.



Stainless steel density refers to substance mass per unit volume, it is one of typical property of stainless steel, commonly, density of stainless steel range from 7600 kg/m3 to 8000 kg/m3.
Stainless steel is a wide used material, containing at least 10.5% of Chromium, and other elements added to form stainless steel structure, these elements have Carbon, Silicon, Manganese, Phosphorus, Sulfur, Nickel, Molybdenum, Titanium and Copper, it is noted as high strength and excellent corrosion resistance.
Densities change depending on these alloy elements changing, different alloy content have different density value, even it is the same grade, it is difficult to calculate accurate density values, theoretic density value is given below for reference.
The following is the comparison of several commonly used stainless steel density, data may not be completely accurate, only for reference.
| Grade | Density ( G / Cm3 ) | Density ( Kg / M3 ) |
|---|---|---|
| 201 202 301 302 303 304 304L 304LN 305 321 |
7.93 | 7930 |
| 309S 310S 316 316L 316Ti 316LN 317 317L 347 |
7.98 | 7980 |
| 904L | 7.98 | 7980 |
| 2205 S31803 |
7.80 | 7800 |
| S32750 | 7.85 | 7850 |
| 403 410 410S 416 431 |
7.75 | 7750 |
| 440A | 7.74 | 7740 |
| 440C | 7.62 | 7620 |
| 420 | 7.73 | 7730 |
| 439 430 430F |
7.70 | 7700 |
| 434 | 7.74 | 7740 |
| 444 | 7.75 | 7750 |
| 405 | 7.72 | 7720 |
*These densities given at standard conditions for temperature and pressure condition.
304 And 316 Stainless Steel Densities
304 and 316 are the most used stainless steel grades, their densities are not the same, this decide by chemical composition and content, stainless steel 304 density is 7930 kg / m3, 316 density is 7980 kg / m3, so when calculating stainless steel weight, it is different between 304 and 316 stainless steel.
Stainless Steel Density Conversion, Kg/M3, G/Cm3 And Lbs/In3
Density of stainless steel calculated by dividing the mass by the volume, usually measured in g/cm3, kg/m3, and lbs/in3, each unit can be converted to other units.
Density Relations With Temperature And Pressure
Densities of stainless steel vary by changing either the temperature or the pressure, in general, increasing the temperature decreases the density, increasing the pressure always increases the density.
Seamless and welded are mainly two types stainless steel tube & stainless steel pipe, there is a debate whether seamless is better than welded for a long time.
Although with improved metallurgy and welding processes in welded tubes, the arguments typically focus on structural integrity and corrosion resistance in weld area.
Obviously, seamless and welded tubes & pipes difference is manufacturing process.
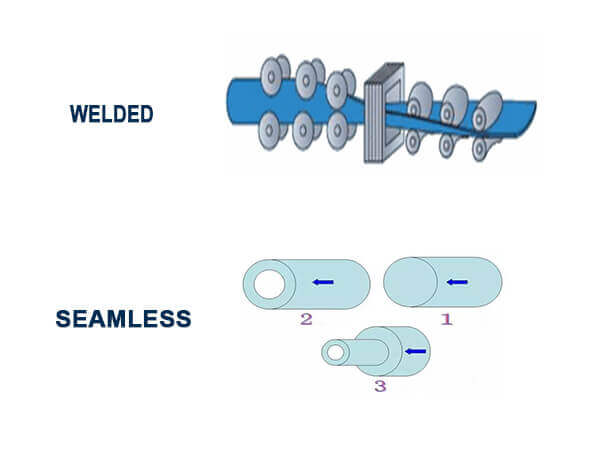
There are some cases where paying extra for a different, higher-performing alloy is necessary.
Depending on the outer dimension, wall thickness and final application, there are different ways of manufacturing welded tubes and pipes, and different manufacturing method.
Welded tube start from stainless steel strip and coil that is passed through grooved rollers till the shape is formed and the free edges are properly shaped for welding, cold forming is performed step by step from flat strip into a round profile, the edges are welded together as they approach the welding rolls.
Typical welding method for strip welded tubes is traditionally autogenous tungsten inert gas (TIG), TIG welding method have some advantages that can effectively protect weld seam.
After welding outside and inside grinding of the weld seam, solution annealing or stress relieving may be necessary by application requirements.

Today, most of the modern welding lines are equipped with inline induction annealing, then straitening and calibration to control dimension of tube & pipe, cut to standard or special lengths, de-burred, NPD & DT testing and inspection should be carried out as standard or requirement.
When large outside diameter or heavy wall or both welded tubes & pipes required, there is a different welding method and process, for large diameter and heavy wall pipes, EFW, ERW and other welding methods can be used.
Starting from stainless steel plate or sheet, the forming is done in a roller bending machine or in a hydraulic press, then some steps are same with strip welded tubes.
For testing and inspection, large diameter sizes, radiography test (RT) and hydrostatic test (HT) are often required.
For large diameter welded pipes, double welding method can be accepted.
Chemical composition inspection, mechanical properties test(tensile strength,yield strength, elongation, flaring, flattening, bending, hardness, impact test), surface and dimension test,no-destructive test, hydrostatic test.
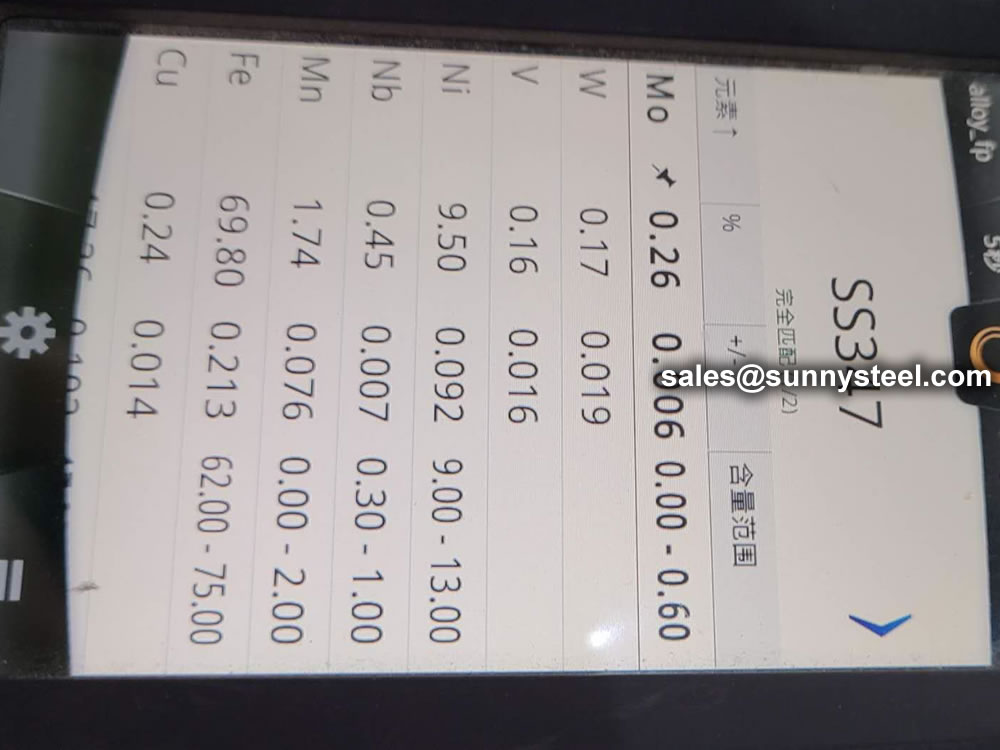
identification of the chemical composition of the metal used to manufacture the fitting. Uses PMI sensors, including X-ray fluorescence or optical emission spectrometry.










Steel pipe delivery status(condition): cold / hard (BK), cold / soft (BKW), after cold stress relief annealing (BKS), annealing (GBK), normalized (NBK).
| Term | Symbol | Explanation |
|---|---|---|
| Cold-finished/hard (cold-finished as-drawn) | BK | No heat treatment after the last cold-forming process. The tubes therefore have only low deformability. |
| Cold-finished/soft (lightly cold-worked) | BKW | After the last heat treatment there is a light finishing pass (cold drawing) With proper subsequent processing, the tube can be cold-formed (e.g. bent, expanded) within certain limits. |
| Annealed | GBK | After the final cold-forming process the tubes are annealed in a controlled atmosphere or under vacuum. |
| Normalized | NBK | The tubes are annealed above the upper transformation point in a controlled atmosphere or under vacuum. |
The general cold strip mills, volume should go through continuous annealing (CAPL unit) to eliminate cold hardening and rolling stress, or batch annealing reach the mechanical properties of the corresponding standard specifies. Cold rolled steel surface quality, appearance, dimensional accuracy better than hot-rolled plate, and right-rolled thin product thickness is about 0.18mm, so the majority of users favor.
Cold rolled steel coil substrate products deep processing of high value-added products. Such as electro-galvanized, hot dip galvanized, electro-galvanized fingerprint resistant, painted steel roll damping composite steel, PVC laminating steel plates, etc., so that the excellent quality of these products has a beautiful, high resistance to corrosion, has been widely used.
Cold rolled steel coil finishing after annealing, cut the head, tail, trimming, flattening, smooth, heavy volume, or longitudinal clipboard. Cold-rolled products are widely used in automobile manufacturing, household electrical appliances, instruments, switches, buildings, office furniture and other industries. Steel plate strapping package weight of 3 to 5 tons. Flat sub-volume typically 3 to 10 tons / volume. Coil diameter 6m.
Bare packing/bundle packing/crate packing/wooden protection at the both sides of tubes and suitably protected for sea-worthly delivery or as requested.
There are probably hundreds of different methods for packing a pipe, and most of them have merit, but there are two principles that are vital for any method to work prevent rusting and Sea transportation security.

Our packing can meet any needs of the customers.












Beveling Stainless Steel tubes contain one end angled, so that it mates with a complementary tube end. Tubes can be beveled for certain applications in order to make welding of stainless steel tubes easier, which saves weld prep time and money compared to hand cutting.
Eastern Steel can heat treat some 400 series stainless steel seamless tube. Heat treating realizes stress as a result of hardening, depending upon the chemistry of the metal being treated. Heat treating is also used for tempering, quenching, normalizing, and solution annealing.
During the honing/turning process, an abrasive rotating tool removes metal from the interior surface of a cylinder, pipe, or tube. The main purpose of honing is to finish the surface to a particular diameter and/or desired cylindrical geometric design. Honing is typically a secondary machining operation that completes a produce/part, relieves stress created during production, or corrects features of the finished product. A typical production cycle is to drill, ream, heat treat, then hone.
POSITIVE MATERIAL IDENTIFICATION (PMI)
In some cases, it may be necessary to complete additional positive material identification testing. Eastern Steel can complete this testing in house or we can work with an independent laboratory to complete the testing. Please contact us for specific requirements.
Ultrasonic Testing (UT) utilizes sound energy at high frequencies, in order to provide examinations and measurements of the stainless steel product. This aids in flaw detection/evaluation, dimensional measurements, etc.
Commonly used alloying elements and their effects are listed in the table given below.
| Alloying Elements | Effect on the Properties |
|---|---|
| Chromium | Increases Resistance to corrosion and oxidation. Increases hardenability and wear resistance. Increases high temperature strength. |
| Nickel | Increases hardenability. Improves toughness. Increases impact strength at low temperatures. |
| Molybdenum | Increases hardenability, high temperature hardness, and wear resistance. Enhances the effects of other alloying elements. Eliminate temper brittleness in steels. Increases high temperature strength. |
| Manganese | Increases hardenability. Combines with sulfur to reduce its adverse effects. |
| Vanadium | Increases hardenability, high temperature hardness, and wear resistance. Improves fatigue resistance. |
| Titanium | Strongest carbide former. Added to stainless steel to prevent precipitation of chromium carbide. |
| Silicon | Removes oxygen in steel making. Improves toughness. Increases hardness ability |
| Boron | Increases hardenability. Produces fine grain size. |
| Aluminum | Forms nitride in nitriding steels. Produces fine grain size in casting. Removes oxygen in steel melting. |
| Cobalt | Increases heat and wear resistance. |
| Tungsten | Increases hardness at elevated temperatures. Refines grain size. |

When you partner with Sunny Steel, you can stop worrying about meeting deadlines thanks to our responsive and timely service. You'll also say goodbye to unnecessary shopping around. Instead, you'll get white glove service from an expert who understands your needs and can get you the materials you need quickly.
